All published articles of this journal are available on ScienceDirect.
Cross-sectional Study on Effects of Ondansetron and Meperidine in the Prevention of Postoperative Shivering after Spinal Anesthesia
Abstract
Purpose:
Post-anesthesia shivering is one of the most common problems after surgery and may lead to multiple complications such as hypoxemia, lactate acidosis and catecholamine release. The purpose of this study was to compare the effects of intravenous meperidine and ondansetron on postoperative shivering in patients having an elective cesarean section under spinal anesthesia.
Design:
Cross-sectional study
Methods:
In this cross-sectional study, 105 patients with the age of 18-45 years and ASA class I-II undergoing elective cesarean section were randomly assigned into three groups. Group O was administered 8mg ondansetron, group M patients were given 0.4mg/kg meperidine and group P was placebo that was administered with 2cc of saline intravenously after the delivery. The incidence and intensity of shivering and postoperative nausea was monitored and recorded by the trained nurse. The data obtained was analyzed using SPSS v18.
Results:
All three groups (n=35) had no significant difference in terms of age, weight and time of spinal anesthesia. Postoperative shivering was reported in 4 patients (11.4%) in group A and 3 patients (8.6%) in group B and in 14 patients (40%) in group C. The incidence of postoperative shivering was significantly less in groups A and B as compared to the placebo, p=0.03. The intensity of shivering was greater in group C as compared to groups A and B, p=0.01. The incidence and intensity of postoperative nausea was significantly less in group A, p=0.03, p<0.001, respectively, while no difference was reported in groups B and C, p<0.05.
Conclusion:
8mg ondansetron and 0.4 mg/kg of meperidine are equally effective in managing postoperative shivering; however, ondansetron has an additional effect of reducing the incidence of postoperative nausea among patients undergoing caesarean section with spinal anesthesia.
1. INTRODUCTION
Spinal anesthesia is commonly used anesthetic technique for a number of surgical procedures including cesarean section due to the rapidity in achieving numbness, clinical efficacy and reduced postoperative complications such as hemodynamic alterations and adverse effects on mental status [1]. Postoperative shivering is one of the common complications associated with spinal anesthesia, reported in about 20-70% of the cases [2]. It is known to slow down the wound healing process, increase oxygen demand and four-fold production of CO2 and hypoxemia, particularly among patients with the history of cardiopulmonary diseases [3, 4]. A number of pharmacological (meperidine, ketamine, tramadol) and non-pharmacological (blankets, bandages, electric and radiant heating, air warming) methodologies have been used to reduce shivering [3]; nonetheless, gold standard technique has not been established so far [5, 6]. Meperidine is one of the most effective anti-shivering agents that acts on thermoregulation centers or opioid receptors to regulate shivering. 0.4mg/kg of meperidine is an optimal dose in the management of postoperative shivering [7, 8].
Evidence has shown the effective of pharmacological agents like clonidine, tramadol and dexmedetomidine for postoperatively shivering [9], however, they are associated with a number of side-effects like bradycardia, prolonged intubation and dry mouth [10].
Serotonin (5-hydroxytryptamine 3 (5-HT3)) is a neurotransmitter that is found in the brain and spinal cord. The 5-HT3 system is known to play a significant role in postoperative shivering [11]. 5-HT3 receptor antagonists such as granisetron have been reported to reduce the incidence of shivering [1]. Ondansetron is also a 5-HT3 antagonist that is widely known for its anti-emetic effects and to treat postoperative nausea and vomit. Some studies have also suggested its efficacy against postoperative shivering [12].
The aim of this study is to evaluate the effects of meperidine and ondansetron on postoperative shivering among patients undergoing elective cesarean section with spinal anesthesia.
2. METHODS
2.1. Study Design and Patient Population
In this double-blinded cross-sectional study, 105 patients aged 18 to 45 years in ASA I and II anesthesia classes, who were referred to Asali Hospital for elective cesarean section (C-section), were randomly assigned to three groups of 35 patients, receiving ondansetron, meperidine or placebo, each. All the patients were provided with the details of the study and written consent for participation was obtained from them. The randomization was performed using a simple randomization method.
Our exclusion criteria included: patients older than 45 years, patients having neurological diseases such as Parkinson’s disease, those with the history of epilepsy, patients allergic to ondansetron or meperidine, history of drug abuse, patients who received blood components during the surgery, history of coronary heart disease, history of hyperthyroidism or hypothyroidism, duration of surgery longer than one hour and patients who received fluid more than 2 liters during the surgery.
This study was approved by the Research Ethics Board of the Lorestan University of Medical Sciences (IR.LUMS.REC.1398.097). https://ethics.research.ac.ir/ProposalCertificateEn.php?id=76654&Print=true&NoPrintHeader=true&NoPrintFooter=true&NoPrintPageBorder=true&LetterPrint=true.
2.2. Anesthesia and Postoperative Care
All the patients received 10 cc/kg of pre-warmed fluid therapy before spinal anesthesia and the temperature of the operation theater and recovery room was maintained at 24-25˚C throughout the course of the study. All the patients (in all three groups) were administered with 12 to 15 mg of isobaric bupivacaine 0.5% at L3-L4 level with needle number 25 as a spinal anesthesia by a specialized and experienced anesthesiologist. Group A received 8 mg of ondansetron, group B received 0.4 mg/kg of meperidine and group C received 2 cc of placebo, intravenously, after the birth of the child. The drugs and placebo were prepared at the same volume and the technician responsible was not included in the study.
Routine care for postoperative shivering was performed in the patients that included using a single covering blanket and oxygen supply by face mask. Intensity of shivering was graded based on a system validated by (0-4 points), where 0 implies no shivering, 1) peripheral vasoconstriction but no visible shivering, 2) shivering seen in one group of muscle, 3) shivering involving two or more groups of muscle and 4) implies whole body shivering. Additionally, postoperative incidence of nausea was also recorded in all the three groups at 12 and 24 postoperative hours and the average was recorded. Intensity of nausea was graded 0-4 points (0: no nausea, 1: mild nausea, 3: moderate nausea, 4: severe nausea. The information was recorded by a trained nurse who was unaware of the type of the drug used for the patient.
2.3. Data Analysis
The data collected was computerized and analyzed using SPSS v18. Mean and standard deviation was used to analyze the data in terms of homogeneity among the groups for parameters including shivering and postoperative nausea using chi-square test. In order to compare quantitative independent variables, Kruskal-Wallis test was used, and if the results were significant for pair wise comparisons, Bonferroni correction was used. The results were reported at a significant level of 0.05.
The work has been reported in line with the STROCSS criteria [13].
3. RESULTS
One hundred and five cesarean section patients were equally divided into three groups of 35 patients each. The mean age (+/-SD) of the patients in groups A, B and C was 30.1 +/- 5.4, 29.51 +/- 5.7 and 29.91 +/- 5.7 years, respectively. The mean age among the 3 groups was not significantly different, p=0.83. Similarly, the mean weight of the patients in the three groups was 78.31 +/- 13.4, 76.94 +/- 8.7 and 76.14 +/- 10.3 kg, respectively, which was also not significantly different, p=0.68. The mean spinal anesthesia time among these three groups was 43.71 +/- 6.5, 42.71 +/- 6.1 and 42.68 +/- 5.8 minutes, respectively, which was not statistically significant, p=0.60.
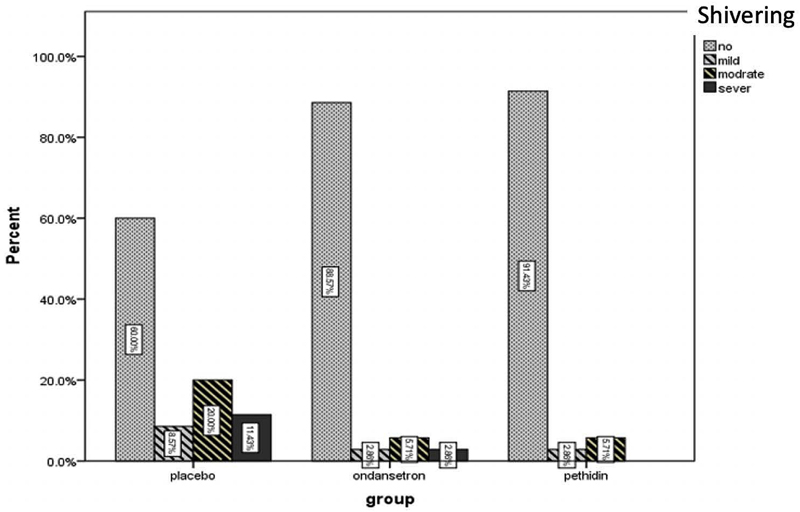
In group A, 4 patients (11.4%) were presented with shivering among which 1 patient (2.9%) had grade two shivering, 2 (5.7%) had grade three and 1 (2.9%) had grade four shivering. In group B, 3 patients (8.6%) were presented with postoperative shivering where 1 patient (2.9%) had grade two and 2 patients (5.7%) had grade three shivering. In placebo group, 14 patients (40%) were presented with shivering after the C-section where 3 patients (8.6%) had grade two, 7 patients (20%) had grade three and 4 (11.4%) had grade four shivering.
There was a significant difference between the three groups in terms of the frequency, p= 0.03, (Table 1), (Fig. 1). The mean intensity of the shivering in groups A, B and C was 0.22 +/- 0.68, 0.34 +/- 0.49 and 1.22 +/- 1.12, respectively. The results from Bonferroni correction showed that the intensity of shivering was significantly lower in groups A and B as compared to group C (placebo), p=0.001, however, shivering intensity was not different in groups A and B, p=0.66, (Table 2), (Fig. 2).
| - | Shivering | Total | p-value | |||
|---|---|---|---|---|---|---|
| Yes | No | |||||
| Group | Placebo | Number | 14 | 21 | 35 | 0.03 |
| Percent | 40 | 60 | 100 | |||
| Ondansetron | Number | 4 | 31 | 35 | ||
| Percent | 11.4 | 88.6 | 100 | |||
| Meperidine | Number | 3 | 32 | 35 | ||
| Percent | 8.6 | 91.4 | 100 | |||
| Total | Number | 21 | 84 | 105 | ||
| Percent | 20 | 80 | 100 | |||
| Group | SD ±Mean | p-value |
|---|---|---|
| Placebo | 1.22±1.12 | <0.001 |
| Ondansetron | 0.22±0.68 | |
| Meperidine | 0.34±0.49 |
| - | Nausea | Total | p-value | |||
|---|---|---|---|---|---|---|
| Yes | No | |||||
| Group | Placebo | Number | 10 | 25 | 35 | 0.03 |
| Percent | 28.6 | 71.4 | 100 | |||
| Ondansetron | Number | 1 | 34 | 35 | ||
| Percent | 2.9 | 97.1 | 100 | |||
| Meperidine | Number | 11 | 24 | 35 | ||
| Percent | 31.4 | 68.6 | 100 | |||
| Total | Number | 21 | 84 | 105 | ||
| Percent | 21 | 79 | 100 | |||
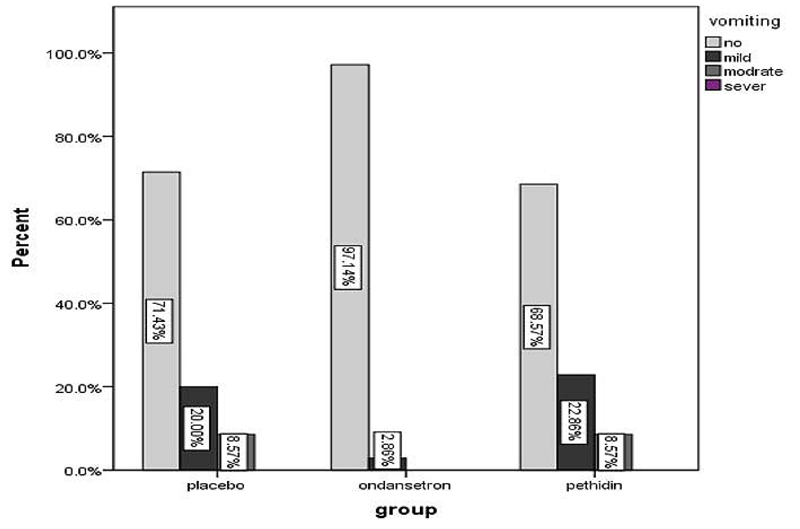
Postoperative nausea was reported in 1 patient (2.9%) in group A, 11 patients (31.4%) in group B and 10 patients (28.6%) in group C, placebo. The difference was significantly different among the three groups, p= 0.03, (Table 3), (Fig. 2). The mean intensity of nausea in groups A, B and C was 0.02 +/- 0.16, 0.4 +/- 0.65 and 0.37 +/- 0.64, respectively. The results from Bonferroni correction showed that the intensity of nausea was significantly lower in group A compared to group B and group C, p<0.001, but not significantly different between group B and group C, (Table 4), (Fig. 2).
Table 4.
| Group | SD ±Mean | p-value |
|---|---|---|
| Placebo | 0.37±0.64 | <0.001 |
| Ondansetron | 0.02±0.16 | |
| Meperidine | 0. 4±0.65 |
4. DISCUSSION
Management of postoperative shivering is an important aspect of patient wellbeing after the surgery, and it is associated with a number of complications [3, 14, 15]. In this study, the effect of ondansetron and meperidine was evaluated in the prevention of postoperative shivering among patients undergoing elective cesarean section, under spinal anesthesia. Our study concluded that the incidence and intensity of postoperative shivering is similar in both the groups and was significantly less as compared to the placebo group. However, the incidence of nausea was significantly less in the ondansetron group.
Internal redistribution of body heat, suppression of thermoregulatory control and loss of heat to the environment in response to local anesthesia contribute chiefly to hypothermia [16]. 5-HT3 antagonist modulates thermoregulation by inhibiting serotonin reuptake by preoptic hypothalamic region [17]. The region is known to contribute to both, heat loss and heat production [18]. The anti-shivering mechanism of meperidine is not fully understood however, Golembiewski, [19], has documented that it is regulated by kappa opioid receptor [12] and/or biogenic monoamine reuptake inhibition, N-methyl-D-aspartate receptor inhibition or activation of alpha-2 adrenoceptors.
A meta-analysis by He et al., including 8 randomized control trials and 905 subjects reported that ondansetron significantly reduces the incidence of post-anesthesia shivering [20]. Other meta-analysis studies also concluded that ondansetron prevents postoperative shivering and the effects of ondansetron and meperidine are similar [8, 9]. In a randomized placebo-controlled trial targeting the comparative effects of ondansetron and meperidine on postoperative shivering following general anesthesia, it is reported that the incidence of shivering in ondansetron and meperidine was significantly lower as compared to the placebo group [21, 22]. The findings from these studies are in parallel to our outcomes. However, the incidence of postoperative nausea was similar in the three groups [23]. In contrast, our study reported reduced incidence of nausea in the ondansetron group compared to meperidine and placebo group. A recent study by Gicheru et al., reported that the weight-adjusted dose of ondansetron at 0.1 mg/kg is more effective in reducing the incidence of postoperative shivering as compared to the fixed dose of 4 mg, among elective C-section patients under spinal anesthesia. Nonetheless, our study is restricted to the fixed dose of 8 mg ondansetron [16], conducted a study comparing two different doses of 4 mg and 8 mg ondansetron and 0.4 mg/kg meperidine after general anesthesia. The results from the study showed that 8 mg of ondansetron was more effective for controlling postoperative shivering than 4 mg ondansetron and similar to 0.4 mg/kg meperidine. Similarly, our study did not report any significant difference among 8 mg ondansetron and 0.4 mg/kg meperidine groups. Their study also concluded that ondansetron groups had significantly lower incidence of postoperative nausea and vomiting, which is similar to our findings.
CONCLUSION
Our study is limited to a small sample size, results presented are the outcomes from the fixed and single dose of ondansetron, we did not obtain the core temperature, and the levels of analgesia were not recorded. Detailed studies including other parameters and variations in the doses can provide a better conclusion and future prospective. 8 mg ondansetron and 0.4 mg/kg meperidine may have similar effects on controlling postoperative shivering; however, ondansetron can have an additional benefit of reducing the incidence of postoperative nausea.
ETHICAL APPROVAL AND CONSENT TO PARTICIPATE
This study was approved by the Research Ethics Board of the Lorestan University of Medical Sciences (IR.LUMS.REC.1398.097). https://ethics.research.ac.ir/ProposalCertificateEn.php?id=76654&Print=true&NoPrintHeader=true&NoPrintFooter=true&NoPrintPageBorder=true&LetterPrint=true
HUMAN AND ANIMAL RIGHTS
All procedures performed in this study involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
CONSENT FOR PUBLICATION
Written consent was obtained from all the participants for the participation in the study.
STANDARDS OF REPORTING
The work has been reported in line with the STROCSS criteria.
AVAILABILITY OF DATA AND MATERIALS
Data sharing is not applicable to this article as no datasets were generated or analyzed during the current study.
FUNDING
None.
CONFLICT OF INTEREST
The authors declare no conflict of interest, financial or otherwise.
ACKNOWLEDGEMENTS
Dr. Sepideh Vahabi: Conceptualized and designed the study, drafted the initial manuscript, and reviewed and revised the manuscript.
Dr. Arash Karimi and Dr. BagherJafari-Takab: Designed the data collection instruments, collected data, carried out the initial analyses, and reviewed and revised the manuscript.
Dr. Siavash Beiranvand: Coordinated and supervised data collection, and critically reviewed the manuscript for important intellectual content.


