All published articles of this journal are available on ScienceDirect.
Effect of Adding Dexmedetomidine to Bupivacaine in Ultrasound Guided Rectus Sheath Block: A Randomized Controlled Double-blinded Study
Abstract
Background:
Extended midline laparotomy incision is accompanied by intense pain postoperatively which affects patients’ physiology; therefore, good control of postoperative pain is mandatory to decrease the adverse effects on the body. Ultrasound-guided Bilateral Rectus Sheath Block (BRSB) is one of the options to achieve this goal.
Objective:
The study aimed to assess the analgesic potency of adding dexmedetomidine to bupivacaine in ultrasound-guided BRSB in cancer patients with a midline laparotomy incision.
Methods:
Sixty adult cancer patients planned for laparotomies with extended midline incision were included. Ultrasound-guided BRSB was performed immediately after the induction of anesthesia.
Patients were classified randomly into two groups; B group, where only bupivacaine was used for BRSB and BD group in whom a mixture of bupivacaine and dexmedetomidine was used.
Results:
A significant decrease in visual analogue scale scores, total morphine consumption, postoperative nausea and vomiting and postoperative cortisol levels was observed in group BD.
Conclusion:
Dexmedetomidine as an adjuvant to bupivacaine in US-guided rectus sheath block bilaterally proved to be effective for proper pain management postoperatively in cancer patients after extended midline abdominal incision.
1. INTRODUCTION
Over the past years, the concept of pain management has extended from simply decreasing pain intensity to optimizing patient’s condition. The goal is to decrease pain scores, stress response that should be avoided in patients, particularly cardiac patients, together with a decrease in analgesics-related adverse effects like nausea, vomiting, retention of urine and over sedation. By achieving these goals, we can certainly facilitate patient recovery and minimize the hospital stay. Improved pain control can be achieved by a combination of different types of regional analgesia with systemic analgesics. The main contributor to pain post abdominal operations is the pain from abdominal wall incision [1].
Many procedures were followed to decrease this intense postoperative pain such as, epidural catheter analgesia, Transverse Abdominis Plane (TAP) block, local wound infiltration, Patient-Controlled Analgesia (PCA), peripheral nerve blocks, in addition to Systemic administration of Non-Steroidal Anti- Inflammatory Drugs (NSAIDs) or opioids [2-4].
The Rectus Sheath Block (RSB) is one of these regional techniques that are used effectively for postoperative analgesia after laparoscopy [5], umbilical hernia repair [6], abdominoplasty [7], upper abdominal [8], and major gynecological surgery [9]. However, the usefulness of traditional local anesthetics to provide analgesia during the course of the postoperative period is restricted by their short duration of action [10]. Dexmedetomidine (DEX) is one of the α -adrenoceptor agonists with α2 -adrenoceptor selectivity [11]. It has been shown as a valuable additive to local anesthetics in neuraxial blocks [12] and peripheral nerve blocks [13] leading to prolongation of postoperative analgesia and better pain control.
The aim of this study was to assess the postoperative analgesic potency of dexmedetomidine as an adjuvant to bupivacaine in ultrasound-guided bilateral rectus sheath block in patients with cancer for major midline laparotomy incision.
2. PATIENTS AND METHODS
This study was conducted between September 2016 and October 2017. The study protocol was approved by the ethical committee of the national cancer institute, Cairo University. An informed consent was taken from each patient included in this study, the night before the day of surgery after explanation to the patients about the possible postoperative complications which include symptoms suggesting local anesthetic toxicity as lightheadedness, dizziness, tinnitus, disorientation, drowsiness, generalized muscle twitching, convulsions, respiratory depression, cardiovascular depression, and collapse, in addition to possible systemic effects of dexmedetomidine, such as bradycardia, hypotension, fainting, and somnolence.
The study involved 60 adult patients (ASA I or II) with abdominal cancer scheduled for midline abdominal operations. After the induction of anesthesia and patient stabilization, the abdomen was sterilized and draped. Then, under complete aseptic conditions, ultrasound-guided Bilateral Rectus Sheath Block (BRSB) was performed . The patients were classified randomly to one of the two studied groups with the help of computer-generated random numbers. In group BD (n = 30), the BRSB was performed by injecting 20 ml of 0.25% bupivacaine in a mixture with 2 µ/kg of dexmedetomidine. In group B (n = 30), 20 ml of 0.25% bupivacaine alone was injected similarly.
The RSB was performed using Short-Axis In-Plane technique. The linear probe with high frequency was placed lateral to the lateral border of rectus abdominus muscle in a transverse plane. The target site for local anesthetic injection was deep to the rectus abdominis muscle, and superficial to the rectus sheath. A single injection was given bilaterally by a 22-gauge, spinal needle. The needle was inserted 3-8 cm lateral to the outer edge of the probe and then advanced from the lateral to the medial. The needle penetrated the lateral border of linea semilunaris and entered the Rectus Abdominis Muscle (RAM). The needle then advanced until it was placed deep to the surface of RAM and the posterior aspect of the rectus sheath. Here, a 1-3 ml of local anesthetic was injected to assure the correct position of the needle by the presence of an anechoic fluid collection. Then, 20 ml of the medication was injected causing the expansion of the anechoic fluid collection. After injection, the transducer was redirected in a cephalad-to-caudad position to confirm spreading inside the posterior rectus sheath compartment. Then same maneuver was done on the other side.
Anesthetic technique was the same for all patients. Midazolam 2 mg IV was administered once to patients in holding area as a premedication. At the operating theatre, monitors including electrocardiogram, pulse oximetry, non-invasive blood pressure and end-tidal carbon dioxide tension (after intubation) were connected. Anesthesia was induced by fentanyl 2 μ/kg, propofol 2 mg/kg, and atracurium 0.5 mg/kg. Patients were ventilated with volume control mode aiming to keep an EtCO2~35 mmHg using O2/air. Anesthesia was maintained using isoflurane and atracurium in increments to keep good level of muscle relaxation. Then the muscle relaxant was reversed by neostigmine 70 µg/kg and atropine 30 µg/kg at the end of the operation. The patients were shifted to the Post-Anesthesia Care Unit (PACU), fully awake and vitally stable and kept for 30 minutes under observation before shifting to the high dependency unit for 24 hours.
The patients were instructed by an anesthetist how to describe their pain on a 10-cm Visual Analogue Scale (VAS) with 0-point means no pain and 10-point means worst pain. Postoperatively, all patients received 1 gm paracetamol infusion/6 hours regularly. An intramuscular morphine sulphate injection 0.1 mg/kg was used as a rescue analgesia if the VAS score was > 5.
The nursing staff and the anesthetist responsible for the collection of data were not aware of group assignment. The data to be collected were VAS score, total number of injection of morphine needed in the 24 hours postoperatively, and the occurrence of nausea and vomiting. Blood samples for cortisol level in the blood were collected while patients were in the holding area before operation and 24 hours post-operatively.
The primary outcome was to assess the intensity of postoperative pain measured by VAS score. The secondary outcomes were postoperative morphine consumption and postoperative cortisol levels.
2.1. Statistical Methods
Statistical analysis was performed using IBM© SPSS© Statistics version 22 (IBM© Corp., Armonk, NY, USA). Numerical data were expressed as mean and standard deviation or median and range as appropriate. Qualitative data were expressed as frequency and percentage. Chi-square test (Fisher’s exact test) was used to examine the relationship between qualitative variables. Comparison between the two groups was made using student t-test or Mann Whitney- U test as appropriate. Comparison between repeated measures was made by paired-test test or Friedman test as appropriate. Bonferroni correction of p values was done for multiple testing. All tests were two-tailed. A p-value < 0.05 was considered statistically significant.
3. RESULTS
There was no significant difference between the two groups regarding age, sex, weight or type of surgery. Table 1 shows the postoperative VAS scores up to 24 hours. The VAS scores of Group BD were significantly lower than that of Group B up to 24 hours postoperatively. The time to the first request of analgesia was significantly longer (p < 0.001) and total postoperative morphine consumption was significantly lower (p < 0.001) in group BD (Table 2). Table 3 shows the number of patients who received morphine during the postoperative period. Also, group BD showed significantly smaller number of patients who developed postoperative nausea and vomiting (Table 4).
| Postoperative VAS score |
Group B (n=30) |
Group BD (n=30) |
p value |
|---|---|---|---|
| After 2 hours | 2 (0-3) | 1 (0-2) | 0.006 |
| After 4 hours | 2 (2-6) | 2 (2-2) | < 0.001 |
| After 6 hours | 5 (3-7) | 4 (2-4) | < 0.001 |
| After 12 hours | 7 (4-8) | 5 (3-5) | < 0.001 |
| After 24 hours | 7 (6-9) | 5 (4-7) | < 0.001 |
|
Group B n=30 |
Group BD n=30 |
p value | |
|---|---|---|---|
| Time to first request of analgesia (hrs) | 6 (4-14) | 15 (4-20) | < 0.001 |
| Total Morphine consumption (mg) | 4 (0-6) | 0 (0-3) | < 0.001 |
|
Group B n=30 |
Group BD n=30 |
p value | |
|---|---|---|---|
| Opioid consumption | – | – | – |
| After 4 hours | 5 (16.7%) | 0 (0.0%) | 0.52 |
| After 6 hours | 10 (33.3%) | 0 (0.0%) | 0.01 |
| After 12 hours | 18 (60.0%) | 0 (0.0%) | < 0.001 |
| After 24 hours | 30 (100.0%) | 7 (23.3%) | < 0.001 |
Table 5 shows that the preoperative serum cortisol levels were comparable in the two groups (p = 0.961). In the two groups, cortisol levels decreased significantly (p < 0.001, for both groups). The postoperative cortisol levels were significantly lower in group BD (p< 0.001). Also, the percentage reduction of serum cortisol was significantly higher in group BD (p < 0.001).
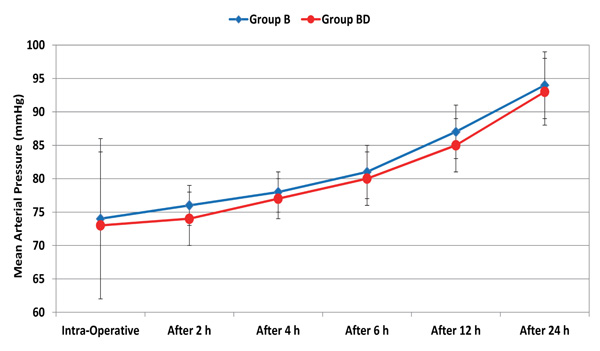
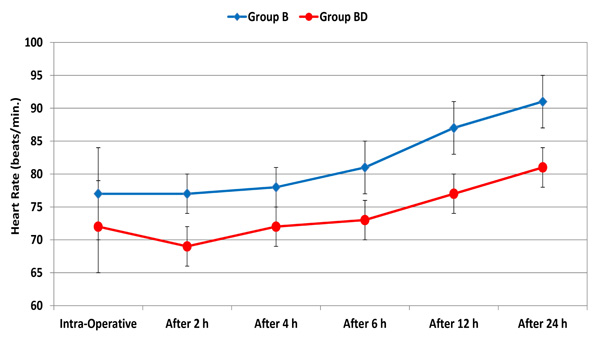
Throughout the 24 postoperative hours, there was no significant difference in mean arterial pressure (MAP) between the two groups. Compared to the intraoperative reading, MAP started to be significantly higher after 12 hours in the two groups (Fig. 1). However, all the values were within the clinically accepted ranges. Starting from 2 hours to 24 hours postoperatively, heart rate was significantly lower in group BD (p < 0.001). Relative to the intraoperative reading, heart rate started to be significantly higher after 12 in the two groups (Fig. 2). However, all values were within the clinically accepted ranges.
| Time |
Group B n=30 |
Group BD n=30 |
p value |
|---|---|---|---|
| After 2 hours | 9 (30.0%) | 0 (0.0%) | 0.02 |
| After 4 hours | 30 (100.0%) | 0 (0.0%) | < 0.001 |
| After 6 hours | 30 (100.0%) | 2 (6.7%) | < 0.001 |
| After 12 hours | 30 (100.0%) | 5 (16.7%) | < 0.001 |
| After 24 hours | 30 (100.0%) | 5 (16.7%) | < 0.001 |
| Serum Cortisol (μg/dL) |
Group B (n=30) |
Group BD (n=30) |
p value |
|---|---|---|---|
| – | – | – | |
| Preoperative | 16.3 ± 1.4 | 16.1 ± 1.4 | 0.691 |
| Postoperative | 14.4 ± 1.8 | 8.6 ± 1.1 | < 0.001 |
| Percentage reduction | -12.9 (-34.2 – 6.5) | -46.7 (-55.4 – 35.9) | < 0.001 |
4. DISCUSSION
This study showed Dexmedetomidine as an adjuvant to bupivacaine in US-guided bilateral rectus sheath block in cancer patients undergoing abdominal surgery with midline incisions associated with a significant reduction in postoperative pain and total morphine consumption with a reduction in the postoperative cortisol level.
Ultrasound-guided BRSB could be more advantageous than the epidural block that is commonly used in cases of extended midline laparotomy incision. It can be a good alternative in cases with absolute contraindications to epidural block as coagulopathy and sepsis. The hemodynamic stability associated with BRSB makes it an acceptable option for hypovolemic patients and patients with cardiovascular diseases. Another major advantage is the absence of the catastrophic complications that may occur with epidural analgesia as epidural hematoma, abscess, or spinal cord injury [14]. Compared with TAP block, another approach of abdominal wall regional anesthesia, BRSB is more effective in midline and paramedian incisions above the umbilicus [15, 16].
Initially, RSB was performed blindly, but now it has been performed using ultrasound guidance [14, 17]; which is, according to our experience, easy, safe and non-time-consuming. The key to success of the regional block is the suitable dose and exact location of the local anesthetic injection. Ultrasound allows proper placement of the LA under vision inside the potential space between posterior rectus sheath and the RAM. This maximizes safety by the prevention of unintentional injection of local anesthesia in the epigastric vessels running in the posterior rectus sheath. In the current study, no signs or symptoms of local anesthetic toxicity in both the groups were recorded.
Dexmedetomidine was found to improve central and peripheral neural blockades by local anesthetics [18, 19]. Several studies have shown that perineural dexmedetomidine as an adjuvant to local anesthetics prolongs the duration of peripheral nerve blocks and provides satisfactory postoperative analgesia [20-23].
To the best of the authors’ knowledge, only one previous study examined the effect of adding dexmedetomidine to bupivacaine during RSB and TAP block in elderly patients during emergency abdominal surgery. DEX prolonged the duration of the sensory blockade and delayed the request of rescue analgesia [24].
In US-guided TAP block for abdominal hysterectomy, DEX was reported to delay the request of rescue analgesia and decrease total analgesic consumption and provided better postoperative pain control [25, 26]. On the contrary, Ding et al. concluded that adding dexmedetomidine does not significantly improve the quality or duration of TAP block [27].
In cases of brachial plexus blockade, the addition of DEX to the local anesthetic provided more rapid onset and longer duration of sensory and motor block with better postoperative analgesia [28]. The same effects were reported when DEX 100 μg was added to bupivacaine during ultrasound-guided combined femoral and sciatic block for below knee surgery [29].
A meta-analysis of 16 randomized controlled trials including 1092 adults was conducted to compare the outcomes between DEX (intrathecal, epidural, or caudal) and bupivacaine or ropivacaine. It decreased pain and prolonged analgesia. Although there was an increased incidence of bradycardia associated with DEX, it was not associated with hypotension and did not warrant treatment [12].
The mechanism of the analgesic action of α2-agonists is probably multifactorial. It may exert an inhibitory neuronal action through the activation of inwardly rectifying G1-protein-gated potassium channels with the subsequent membrane hyperpolarization and a decrease of firing rate of CNS excitable cells [30]. Another action advocated is the reduction of calcium transfer into the cells inhibiting neurotransmitter release. This action is mediated through N-type voltage-gated calcium channels, is independent of cAMP and protein phosphorylation and is mediated by G0 proteins [31]. It can delay the absorption and improve the tissue distribution of local anesthetic due to vasoconstriction around the site of injection [32, 33]. Another theory is the direct effect on peripheral nerve activity by blocking the Ih current. This current acts to reset the nerve from a hyperpolarized state back to the resting membrane potential [34]. If the nerve is kept in a hyperpolarized state, it will be unable to generate a new action potential [35].
CONCLUSION
The addition of dexmedetomidine 2 μg/kg to bupivacaine 0.25% in the bilateral rectus sheath block is an effective modality for the management of postoperative pain in cancer patients undergoing midline abdominal surgery under general anesthesia and with no undesirable side effect. Dexmedetomidine injection into the rectus sheath reduced postoperative morphine consumption and decreased the postoperative cortisol level. Lastly, ultrasound as guidance for rectus sheath block was easy and safe practice.
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
The study was approved by the Local Ethical Committee. The study implemented the principles of the Declaration of Helsinki (1964) and its following revisions. One day before surgery, the study protocol was discussed with all the patients with complete explanation of the procedure and its possible adverse effects.
HUMAN AND ANIMAL RIGHTS
No Animals were used in this research. All human research procedures followed were in accordance with the ethical standards of the committee responsible for human experimentation (institutional and national), and with the Helsinki Declaration of 1975, as revised in 2013.
CONSENT FOR PUBLICATION
Written informed consent was provided by each participant.
AVAILABILITY OF DATA AND MATERIALS
Not applicable.
FUNDING
None.
CONFLICT OF INTEREST
The authors declare no conflict of interest, financial or otherwise.
ACKNOWLEDGEMENTS
Declared none.


