All published articles of this journal are available on ScienceDirect.
Intraoperative Dexmedetomidine has no Effect on Postoperative Pain Scores for Posterior Spinal Fusion
Abstract
Background:
Posterior Spinal Fusion (PSF) for idiopathic scoliosis results in severe postoperative pain. At our institution, a protocol for postoperative analgesia is followed, but anesthetic maintenance is decided by the anesthesiologist. Previous studies have shown that postoperative use of dexmedetomidine may improve analgesia for these patients, but the effect of intraoperative dexmedetomidine on postoperative pain scores remains unknown.
Purpose:
We sought to retrospectively compare pain scores from the Postoperative Anesthesia Care Unit (PACU) and from PACU discharge until midnight between PSF patients who did and did not receive intraoperative dexmedetomidine.
Methods:
After obtaining IRB approval, we retrospectively identified 79 patients aged 10-17 years who had undergone PSF for idiopathic scoliosis from June 2015-August 2018 and who received intrathecal morphine. Patients were then divided into two groups based on whether or not they received intraoperative dexmedetomidine. A multivariable linear regression model was constructed with the dependent variable of highest PACU pain score and exposure of interest intraoperative dexmedetomidine use. Secondary analyses were conducted similarly within those who received dexmedetomidine to examine the effects of dose on PACU pain scores, using a p-value < 0.05.
Results:
After adjusting for age, weight, sex, levels fused, intrathecal morphine, diazepam, and ketamine doses, there was no statistically significant difference in average PACU pain scores between those who did and did not receive intraoperative dexmedetomidine (β = -0.85, 95% CI: -2.48, 0.68; p = 0.31).
Conclusion:
Intraoperative use of dexmedetomidine during posterior spinal fusion for adolescent idiopathic scoliosis appears to have no effect on postoperative pain scores.
1. INTRODUCTION
Posterior Spinal Fusion (PSF) to correct adolescent idiopathic scoliosis is known to be associated with severe postoperative pain [1-8]. Commonly used techniques for postoperative analgesia include a single preoperative injection of intrathecal morphine [1-3], Patient Controlled Analgesia (PCA) [4, 5], and epidural [6-8]. There is also literature to support the use of dexmedetomidine postoperatively to improve analgesia for these patients [9-11]. Non-narcotic adjuvant medications commonly used during the perioperative period include acetaminophen, ketorolac, and diazepam. While a wide range of acceptable anesthetic techniques have been described in the literature, Total Intravenous Anesthesia (TIVA) with propofol and remifentanil is used most commonly. Propofol is frequently chosen over the use of dexmedetomidine for intraoperative maintenance of anesthesia due to its lower effect on transcranial motor evoked potential signals [12]. However, some anesthesiologists choose to use dexmedetomidine for intraoperative maintenance of anesthesia due to its potential for decreased time to extubation relative to propofol [13].
Dexmedetomidine, an α-2 adrenoreceptor agonist used primarily as a sedative, was approved for clinical use in 1999 (https://www.accessdata.fda.gov/drugsatfda_docs/label/2013/021038s021lbl.pdf). Its pharmacologic effects include decreased sympathetic tone, reduction in anesthetic and narcotic requirements, analgesia, and decreased neuroendocrine and hemodynamic effects from surgery. It is frequently used as an adjuvant to general anesthesia and as an additive to neuraxial and regional anesthetic techniques. Recent literature [9, 10,14] has also shown a benefit ofperioperative use of dexmedetomidine to improve analgesia for such patients, though whether or not intraoperative use of dexmedetomidine in the setting of intrathecal morphine has any effect on postoperative pain scores remains unknown.
In our institution, patients have routinely received intrathecal morphine for analgesia for PSF surgery since June 2014. There is no standardized intraoperative anesthetic at our institution for PSF, resulting in about half of our anesthesiologists using propofol/remifentanil/isoflurane for maintenance of anesthesia and the other half choosing dexmedetomidine/remifentanil/isoflurane. In this retrospective review, we sought to compare postoperative pain scores for those undergoing PSF between those with and without intraoperative administration of dexmedetomidine. We hypothesized that dexmedetomidine would decrease postoperative pain scores in the PACU but would also increase PACU length of stay.
2. METHODS
After obtaining approval of the Institutional Review Board (HUM00094624, AME 00082368), we retrospectively identified patients aged 10-17 years in our electronic database who had undergone PSF for Adolescent Idiopathic Scoliosis (AIS) between June 2015 and August 2018. Patients were excluded from the analysis if they had non-idiopathic scoliosis, had a designation of American Society of Anesthesiology (ASA) physical status 4, had a previous spinal fusion, or if they did not receive intrathecal morphine.
Institutional protocol for postoperative analgesia dictates that each of these patients receive 8-10 mcg/kg of intrathecal morphine after induction of general anesthesia [1]. Additionally, all patients were scheduled to receive 0.1 mg/kg of oral oxycodone (maximum 5 mg) 16 hours after the injection of intrathecal morphine with no additional intravenous narcotics ordered. Standard adjuvant pain medications (acetaminophen, ketorolac, and diazepam) and medications for adverse events (ondansetron, nalbuphine, naloxone) were ordered at appropriate weight-based doses for all patients in both groups. All patients were recovered in the PACU prior to being discharged to the general care floor. Pain was rated by patients on a numerical 0-10 scale and sedation was scored on the 0-4 University of Michigan Sedation Scale (UMSS) [15].
A trained research assistant reviewed the medical records to collect patient data, including demographics age, weight, and gender; surgical and anesthesia characteristics including levels fused, estimated blood loss, intraoperative medications administered; pain and sedation scores in the PACU and from PACU discharge until midnight; any adverse event, defined as the inability to extubate, reintubation, and need for naloxone; any administration of analgesics, antiemetics and antipruritics; and time to PACU discharge. The highest and lowest pain scores were recorded in the PACU and from PACU discharge to midnight on the day of surgery.
The primary outcome for this study was the highest pain score recorded in the PACU. The exposure of interest was intraoperative use of dexmedetomidine, defined as any use of dexmedetomidine from the time the anesthesiologist began caring for the patient until the patient arrived in the PACU. When dexmedetomidine is used intraoperatively for these cases, it is as an infusion with or without a bolus/loading dose. The infusion rates typically range from 0.1-0.5 mcg/kg/hr, with a max of 1.0 mcg/kg/hr. When dexmedetomidine is not used, a propofol infusion, titrated to sufficient anesthetic depth, is most commonly used instead. Alternative techniques include the use of ketamine infusions or remifentanil infusions without either propofol or dexmedetomidine.
2.1. Statistical Analysis
Descriptive statistics were presented as frequencies with percentages or medians with 25th and 75th percentiles. The normality of continuous data was assessed using histograms and the Shapiro-Wilks test. Univariate comparisons between those who did and did not receive dexmedetomidine were conducted using Chi-square or Fisher’s exact tests for categorical variables and with Wilcoxon rank-sum tests for continuous variables, as appropriate.
To determine if dexmedetomidine use was associated with a decrease in the highest PACU pain score, a multivariable linear regression model was constructed with the dependent variable of highest PACU pain score and variable of interest of intraoperative dexmedetomidine use. Clinically significant covariates chosen for model inclusion were age, weight (kg), sex, intrathecal morphine dose (mcg/kg), levels of the spine fused, diazepam dose (mg/kg), ketamine dose (mg/kg), prior inclusion in a gabapentin study, and an interaction between prior inclusion in a gabapentin study with intrathecal morphine dose (mcg/kg). Prior to model construction, collinearity between covariates was assessed using Pearson correlation coefficients, and a correlation of > = 0.70 was deemed to indicate significant collinearity. If significant collinearity was detected, the variable with the larger univariate effect size would be selected to remain in the model; however, no significant collinearity was detected among the covariates. To account for overfitting of the model based on the 10:1 rule, 1000-sample bootstrap-adjusted 95% confidence intervals and p-values were reported. Measures of effect for the exposure of interest were reported as model beta coefficient with standard error, representing the mean difference in the highest PACU pain score for those in the dexmedetomidine group vs. those who did not receive dexmedetomidine after adjusting for the factors specified above. A sensitivity analysis was conducted on the same model, excluding those who received intraoperative ketamine.
Secondary analyses were conducted similarly within those who received dexmedetomidine to examine the effects of the dose of dexmedetomidine on PACU pain scores. For the secondary analyses, measures of effect were reported as model beta coefficient with standard error, representing the change in mean highest PACU pain score for every one-unit increase in the dose of dexmedetomidine given after adjusting for the factors specified above. To account for overfitting of the model based on the 10:1 rule, 1000-sample bootstrap-adjusted 95% confidence intervals and p-values were reported.
A p-value of 0.05 was considered statistically significant, and all analyses conducted were two-sided. All analyses were conducted using SAS v. 9.4 (SAS Institute, Cary, NC) and SPSS v. 25 (IBM). The full analytic plan was developed post-hoc.
2.2. Power Analysis
Due to the retrospective nature of the study, an a priori power calculation was not conducted. Based on a clinically significant difference in the highest PACU pain score between groups of 2, and a sample standard deviation of 3.0 for the highest PACU pain score, 39 subjects in both the dexmedetomidine and no dexmedetomidine groups (total n = 78) are needed to provide 81% power to detect this difference using a Wilcoxon rank-sum test with an alpha of 0.05. The sample size was computed using PASS 2019 (PASS 2019 Power Analysis and Sample Size Software (2019). NCSS, LLC. Kaysville, Utah, USA, ncss.com/software/pass).
3. RESULTS
During the study period, we identified 40 patients who received propofol as part of their anesthetic maintenance following intrathecal morphine for PSF and 39 patients who received dexmedetomidine as part of their anesthetic maintenance following intrathecal morphine for PSF. Patients in the dexmedetomidine group received a mean total dose of 1.3 µg/kg (range 1.0-1.5 µg/kg), with only 2/39 patients (5%) receiving a dexmedetomidine bolus (10 µg and 12 µg respectively).
There were no statistically significant differences between demographic and surgical characteristics between those who did and did not receive dexmedetomidine (Tables 1 and 2). Patients in the dexmedetomidine group received significantly more intrathecal morphine (median 10.0, IQR 8.0-12.0) than those not in the dexmedetomidine group (median 6.2, IQR 5.9-10.0, p = 0.01). Additionally, those in the dexmedetomidine group had a significantly shorter PACU length of stay (median 1.9 hours, IQR 1.5-2.5 vs. median 2.3, IQR 2.0-3.2; p = 0.01).
After adjusting for age, weight, intrathecal morphine dose, levels fused, diazepam dose, ketamine dose, sex, prior inclusion in a gabapentin study, and the interaction between prior inclusion in a gabapentin study and intrathecal morphine dose, there was no statistically significant difference in average PACU pain scores (incorporating both lowest and highest scores) between those who did and did not receive intraoperative dexmedetomidine (β = -0.85, 95% CI: -2.48 to 0.68; p = 0.31; Table 3). The same results were seen after excluding those who received ketamine infusions (β = -0.68, 95% CI: -2.56 to 1.11; p = 0.47).
| - | No Dexmedetomidine (N = 40) |
Dexmedetomidine (N = 39) |
P-Value |
| Age | 14.4 [13.1 to 15.9] | 14.4 [13.0 to 15.4] | 0.57 |
| Weight (kg) | 55.7 [44.9 to 65.1] | 55.0 [48.0 to 68.1] | 0.44 |
| Height (cm) | 160.0 [155.6 to 165.1] | 162.6 [158.8 to 167.6] | 0.26 |
| BMI | 21.4 [18.6 to 24.5] | 20.7 [18.3 to 26.1] | 0.75 |
| Female Sex | 33 (82.5) | 32 (82.1) | 0.96 |
| Prior inclusion in gabapentin study | 22 (55.0) | 6 (15.4) | <0.001 |
| - | No Dexmedetomidine (N = 40) | Dexmedetomidine (N = 39) | P-Value |
| Intrathecal Morphine Dose (mg/kg) | 6.2 [5.9 to 10.0] | 10.0 [8.0 to 10.2] | 0.01 |
| Levels Fused | 10.0 [8.5 to 11.0] | 10.0 [8.0 to 11.0] | 0.91 |
| Ketamine Use | 14 (35.0) | 7 (18.0) | 0.09 |
| Ketamine Dose (mg/kg) | 1.1 [0.5 to 2.8] | 1.0 [0.5 to 1.1] | 0.33 |
| Diazepam Use | 20 (50.0) | 17 (43.6) | 0.57 |
| Diazepam Dose (mg/kg) | 0.1 [0.0 to 0.1] | 0.1 [0.0 to 0.1] | 0.55 |
Within those who received dexmedetomidine, after adjusting for age, weight, intrathecal morphine dose, levels fused, diazepam dose, ketamine dose, sex, prior inclusion in a gabapentin study, and the interaction between prior inclusion in a gabapentin study and intrathecal morphine dose, there was no statistically significant change in average PACU pain scores as dexmedetomidine dose increased (β = -1.11, 95% CI: -3.81, 1.56; p = 0.41). Similar results were seen after excluding ketamine infusion patients (β = -1.76, 95% CI: -4.88, 1.74; p = 0.31). There were no statistically significant associations in either the univariate or multivariate analyses for the outcome of PACU pain score, and thus the results were consistent. Likewise, there were no statistically significant associations in our sensitivity analyses excluding those who received ketamine, suggesting that the use of ketamine, even in a fairly significant portion of our population, did not skew our results.
| - | No Dexmedetomidine (N = 40) | Dexmedetomidine (N = 39) | P-Value |
| PACU Length of Stay (hours) | 2.3 [2.0 to 3.2] | 1.9 [1.5 to 2.5] | 0.01 |
| PACU Morphine Use | 1 (2.5) | 2 (5.1) | 0.61 |
| Highest PACU Pain | 5.0 [3.0 to 6.5] | 3.0 [0.0 to 6.0] | 0.12 |
| Highest PACU Pain > 4 | 23 (57.5) | 15 (38.5) | 0.12 |
| Lowest PACU Pain | 2.0 [0.0 to 3.0] | 1.0 [0.0 to 3.0] | 0.64 |
| Highest Pain PACU End - Midnight | 3.0 [0.0 to 7.0] | 2.0 [0.0 to 5.0] | 0.09 |
| Lowest Pain PACU End-Midnight | 0.0 [0.0 to 3.5] | 0.5 [0.0 to 3.0] | 0.87 |
4. DISCUSSION
We hypothesized that dexmedetomidine would decrease postoperative pain scores in the PACU but would also increase PACU length of stay. We predicted this outcome because of the well-known analgesic effects of dexmedetomidine [10, 11, 14,16,17] combined with its relatively prolonged context-sensitive half-life [18]. Dexmedetomidine has recently come into favor for its analgesic properties in other types of surgeries that are also known to be painful, such as pediatric tonsillectomies [16, 17]. While a few other institutions have used dexmedetomidine infusions postoperatively for analgesia following PSF, we have not instituted that protocol at our institution as doing so would require Pediatric Intensive Care Unit (PICU) admission, which would significantly drive up costs and decrease PICU bed availability for other patients. At our institution, nearly all patients who undergo PSF for idiopathic scoliosis recover on the general care ward and are discharged on post-operative day (POD) 2 or 3 [1]. Therefore, in order to continue to have these patients recover on the general care ward, we wanted to see if the analgesic effects of intraoperative dexmedetomidine might result in improved postoperative analgesia. Unfortunately, while there was a trend toward lower mean postoperative pain scores in patients who received dexmedetomidine, the difference was not statistically significant. Furthermore, neither sensitivity nor secondary analyses showed any difference, thereby suggesting that there is no association between the use of intraoperative dexmedetomidine and postoperative pain scores. The use of ketamine infusions for a significant number of our patients, especially in the no dexmedetomidine group, impacted the amount of intrathecal morphine that these patients received, but secondary analyses did not indicate that this had any impact on pain scores. While most of our anesthesiologists use a combination of propofol / remifentanil / isoflurane or dexmedetomidine / remifentanil / isoflurane for maintenance of anesthesia in these cases, there is at least one anesthesiologist who uses a ketamine infusion along with dexmedetomidine as their preferred method. As ketamine has its own profound analgesic properties, when it is used, the dose of intrathecal morphine is decreased to 4-6 mcg/kg. Given this change, we did secondary analyses to attempt to delineate if there was any impact; it appeared that there was not, further strengthening our finding that intraoperative use of dexmedetomidine appeared to have no impact on postoperative pain scores.
Contrary to our hypothesis, the dexmedetomidine group showed a trend toward earlier PACU discharge compared to the no dexmedetomidine group. This was surprising as one of the most common concerns voiced by surgeons and anesthesiologists about the use of dexmedetomidine is that it may prolong PACU length of stay [18]. It may actually be that for these cases, given their length and the fact that rarely was a dexmedetomidine bolus dose used, the context-sensitive half-life of propofol is longer compared to that for dexmedetomidine [19, 20]. If a prospective study could show a significantly shorter time to PACU discharge following the use of intraoperative dexmedetomidine for PSF cases, this may be a reason to more strongly advocate for its use, as a disproportionate number of these cases tend to be done during the busy summer months when PACU is routinely operating at maximum capacity.
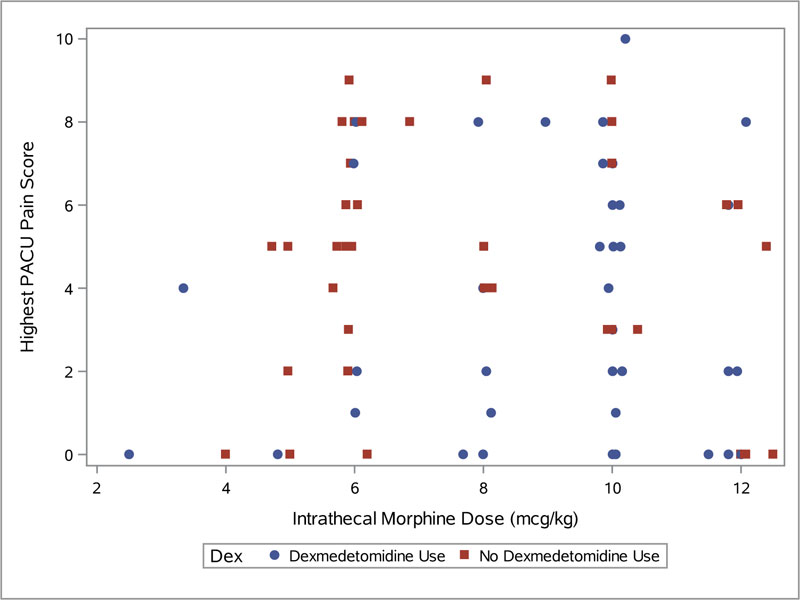
Additionally, it was interesting to see that intrathecal morphine dose appears to have no impact on pain scores in the PACU, independent of whether or not dexmedetomidine was used (Fig. 1). Further studies, possibly prospective, may be indicated to examine this relationship in more detail.
Our institution routinely uses a multidisciplinary clinical care protocol following PSF for idiopathic scoliosis, which guides analgesic therapy, wound care, physical therapy, goals, and diet advancement in the postoperative period. As a result of this and improved postoperative analgesia, our patients are routinely discharged on POD 2 or 3, with some patients occasionally discharged on POD 1. So far, we have not attempted to standardize any aspect of the intraoperative anesthetic beyond the use of intrathecal morphine. Despite a strong push nationwide in hospitals for Enhanced Recovery after Surgery (ERAS) protocols for many complex surgeries, which sometimes extend into an intraoperative protocol, we cannot advocate for or against the intraoperative use of dexmedetomidine on the basis of improved postoperative analgesia. It appears based on our data that standardizing an anesthetic technique for maintenance during these complex cases, which require a multimodal technique due to the necessity of neuromonitoring of both somatosensory evoked potentials and transcranial motor evoked potentials, will not impact postoperative pain. As inadequate pain control remains the rate limiting factor for hospital discharge, it is thus highly unlikely that the use of dexmedetomidine intraoperatively would have any impact on the length of stay after PSF surgery.
This study has numerous strengths, most significant being that despite performing multiple secondary analyses, we were still unable to detect an effect of intraoperative dexmedetomidine on postoperative pain scores, strengthening the argument that there is not one. As a retrospective study, one of the major limitations of this study is that the data collected is subject to availability based on completeness of clinical reporting. Additionally, we were not able to standardize intraoperative medications, resulting in variability in dosing.
CONCLUSION
Intraoperative use of dexmedetomidine for posterior spinal fusion in adolescent idiopathic scoliosis does not appear to be associated with decreased postoperative pain scores.
IMPLICATION STATEMENT
In a retrospective review of intraoperative use of dexmedetomidine for posterior spinal fusion in adolescent idiopathic scoliosis, there was no effect on postoperative pain scores but a trend toward faster readiness for discharge from the postanesthesia care unit was observed.
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
Ethical approval granted by the University of Michigan IRB, USA (study number HUM 00094642, amendment 00082368, approved September 7, 2018).
HUMAN AND ANIMAL RIGHTS
No animals were used in this research. All human research procedures followed were in accordance with the ethical standards of the committee responsible for human experimentation (institutional and national), and with the Helsinki Declaration of 1975, as revised in 2013.
CONSENT FOR PUBLICATION
All patients participated on a voluntary basis and gave their informed consent.
AVAILABILITY OF DATA AND MATERIALS
Not applicable.
FUNDING
None.
CONFLICT OF INTEREST
The authors declare no conflict of interest, financial or otherwise.
ACKNOWLEDGEMENTS
Michelle Burke and Jennylee Swallow, both assisted with data collection.


